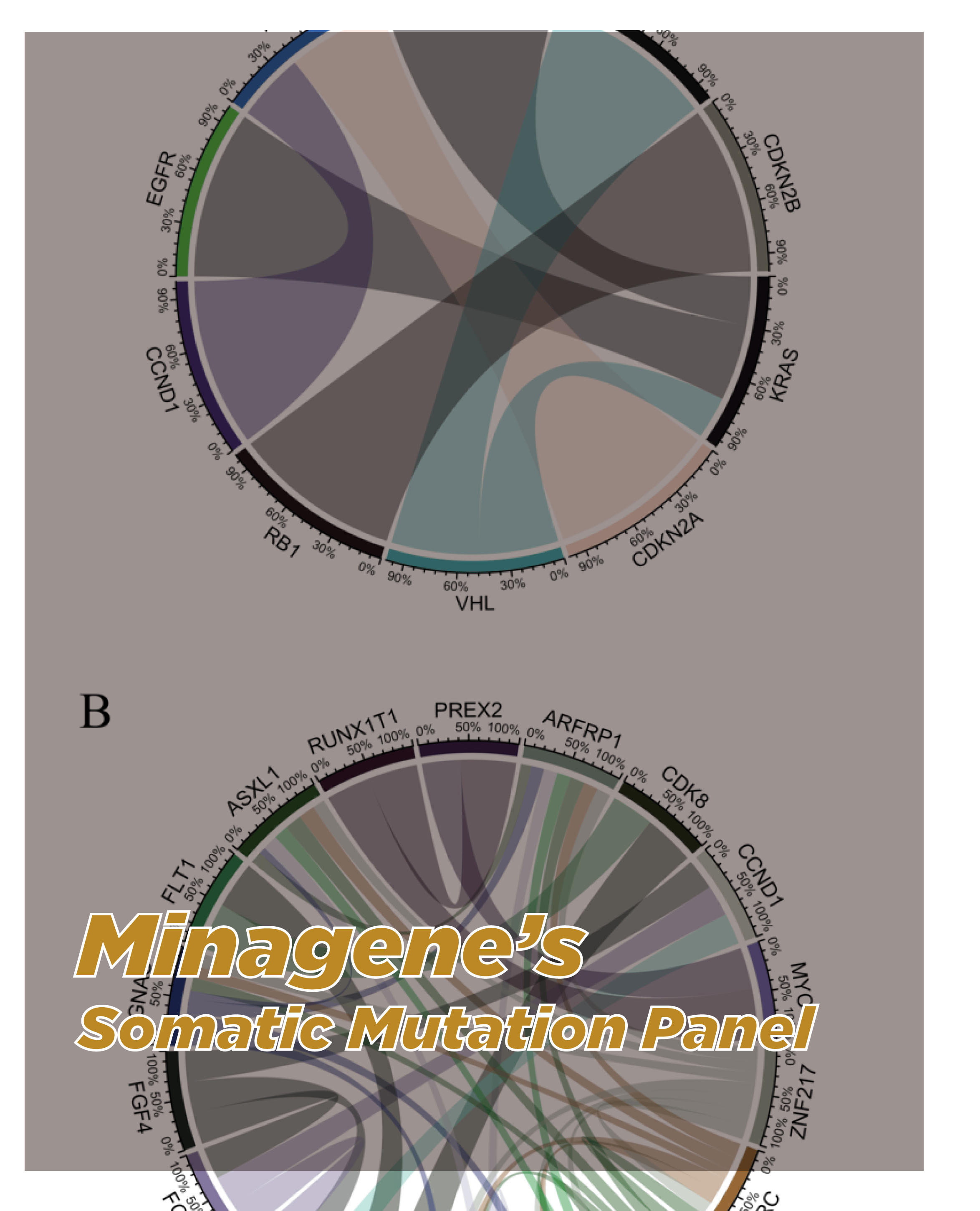
Overview
Minagene's Tumor Mutation Burden (TMB) Test is a specialized genetic test designed to assess the genomic instability of tumors by measuring the tumor mutation burden. This advanced test utilizes high-throughput sequencing techniques to analyze the number of somatic mutations present in the tumor genome, providing valuable insights into tumor biology, prognosis, and potential response to immunotherapies.
Methodology
The TMB Test employs next-generation sequencing (NGS) technology to analyze the tumor genome and detect somatic mutations. It assesses the total number of non-synonymous somatic mutations, including base substitutions, insertions, and deletions, across a broad range of genes. By quantifying the mutation burden, the test provides a measure of genomic instability and potential neoantigen formation in the tumor.
Key Highlights of the Test:
-Comprehensive Genomic Instability Assessment: Minagene’s TMB Test offers a comprehensive analysis of genomic instability by quantifying the tumor mutation burden. It assesses the total number of somatic mutations, including both high and low allele frequency variants, providing a more comprehensive understanding of the tumor’s genetic landscape.
-Prognostic and Predictive Value: TMB has emerged as a valuable biomarker with prognostic and predictive value in cancer. High TMB has been associated with increased response rates to immune checkpoint inhibitors and improved survival outcomes in certain cancer types. The TMB Test helps identify patients who may benefit from immunotherapy and informs treatment decisions.
-Prediction of Immunotherapy Response: TMB is recognized as a potential biomarker for predicting response to immune checkpoint inhibitors, such as PD-1/PD-L1 inhibitors. The TMB Test provides valuable information on the tumor’s mutational load, enabling clinicians to identify patients with a higher likelihood of benefiting from immunotherapeutic approaches.
-Complementary Biomarker: TMB assessment complements other biomarkers, such as PD-L1 expression, in guiding immunotherapy treatment decisions. The combination of TMB and PD-L1 expression analysis provides a more comprehensive understanding of a patient’s potential response to immune checkpoint inhibitors.
-Research and Clinical Trials: The TMB Test can be used in research studies and clinical trials focusing on cancer. The test’s comprehensive analysis of the tumor genome and assessment of genomic instability contribute to a better understanding of tumor biology and the development of targeted therapies.
-Treatment Decision-Making: The TMB Test is considered when making treatment decisions, especially for patients with advanced or metastatic cancer. It helps identify patients who may benefit from immunotherapy, particularly immune checkpoint inhibitors, by predicting potential treatment response based on the tumor’s mutation burden.
-Prognostic Assessment: The test aids in prognostic assessment by evaluating the tumor’s genomic instability. High TMB has been associated with improved survival outcomes in certain cancer types, providing valuable prognostic information for patient management.
-Immunotherapy Response Prediction: The TMB Test is valuable for predicting response to immune checkpoint inhibitors. Patients with high TMB are more likely to respond to immunotherapeutic approaches, making the test useful in guiding treatment decisions and optimizing patient outcomes.
-Combination Biomarker Analysis: TMB assessment complements other biomarkers, such as PD-L1 expression, in guiding immunotherapy treatment decisions. Combined analysis of TMB and PD-L1 expression provides a more comprehensive understanding of a patient’s potential response to immune checkpoint inhibitors.
-Research and Clinical Trials: The TMB Test can be utilized in research studies and clinical trials focusing on cancer. The comprehensive genomic analysis provided by the test contributes to a better understanding of tumor biology, treatment response, and the development of personalized therapies.